Abstract
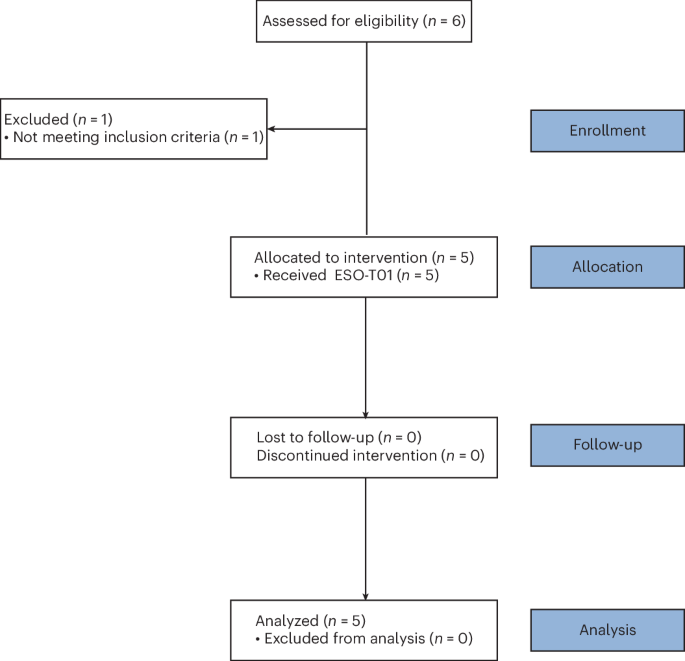
In vivo chimeric antigen receptor (CAR)-T cell generation can bypass ex vivo manufacturing and lymphodepletion, potentially simplifying and accelerating access to cellular therapy; preliminary clinical experience supports feasibility and suggests preliminary efficacy. This phase 1, single-arm, open-label trial evaluated the safety and tolerability of ESO-T01, a nanobody-directed, immune-s...
This study presents an innovative approach to CAR-T cell therapy for RRMM, which has significant potential due to its avoidance of apheresis and ex vivo expansion. However, it is essential to consider the limitations of this small-scale phase 1 trial, including the need for further studies to assess long-term safety and efficacy. Additionally, it's important to examine the broader implications of this technology on the accessibility and affordability of CAR-T cell therapies.
Patterns detected: A...